Strain‐Driven Dyotropic Rearrangement: A Unified Ring‐Expansion Approach to α‐Methylene‐γ‐butyrolactones - Lei - 2021 - Angewandte Chemie International Edition - Wiley Online Library
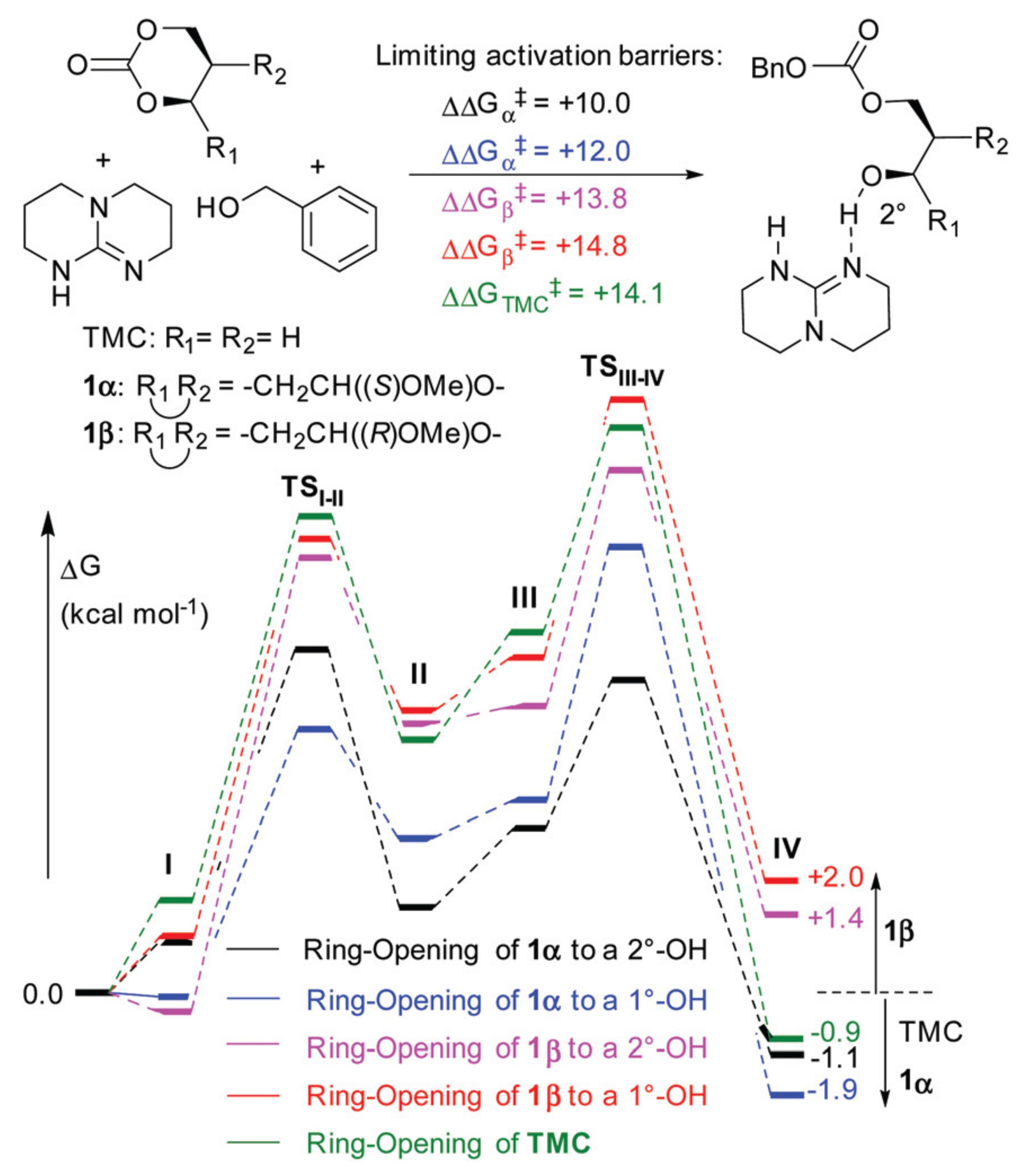
Polymers | Free Full-Text | DFT Modeling of Organocatalytic Ring-Opening Polymerization of Cyclic Esters: A Crucial Role of Proton Exchange and Hydrogen Bonding | HTML

Oxidative opening of the aromatic ring: Tracing the natural history of a large superfamily of dioxygenase domains and their relatives - Journal of Biological Chemistry

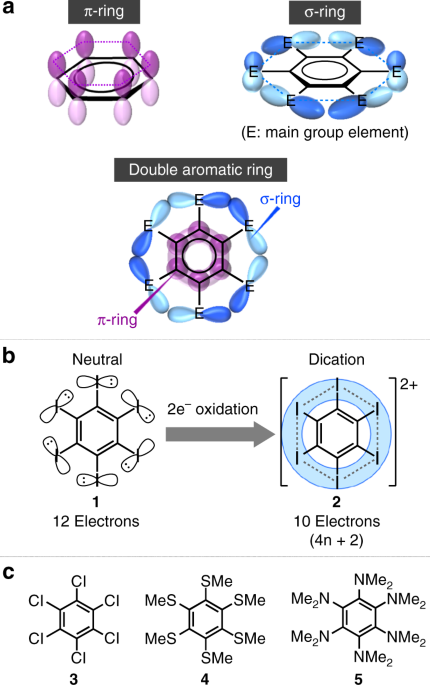
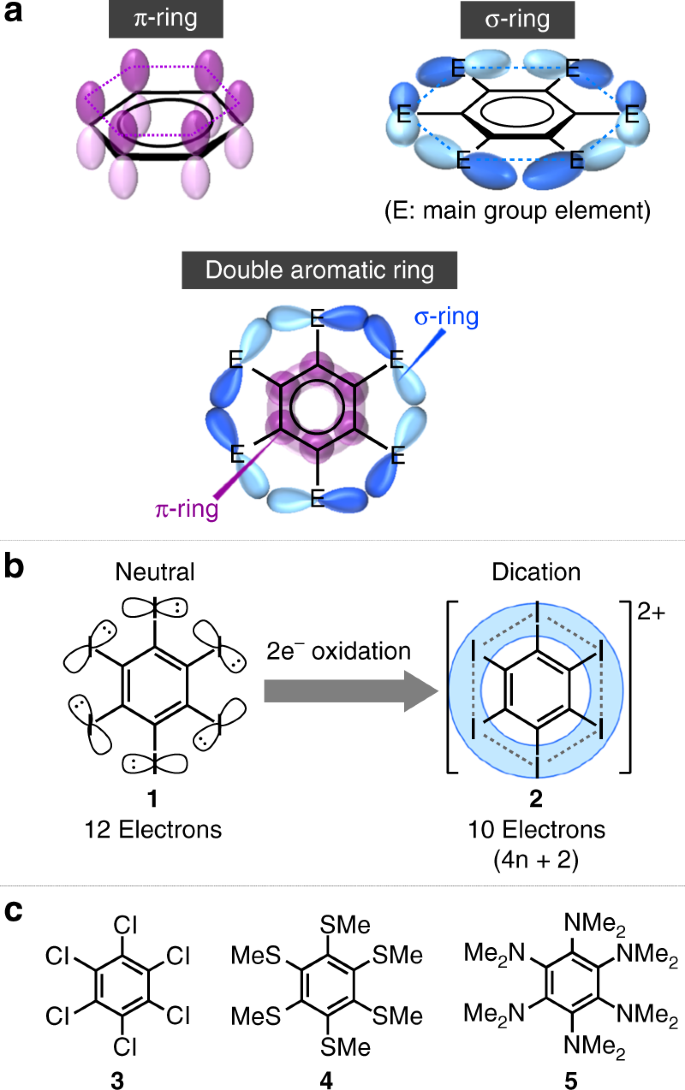
Inorganics | Free Full-Text | Reactivity Studies of Phosphinines: The Selenation of Diphenyl-Phosphine Substituents and Formation of a Chelating Bis(Phosphinine) Palladium(II) Complex | HTML
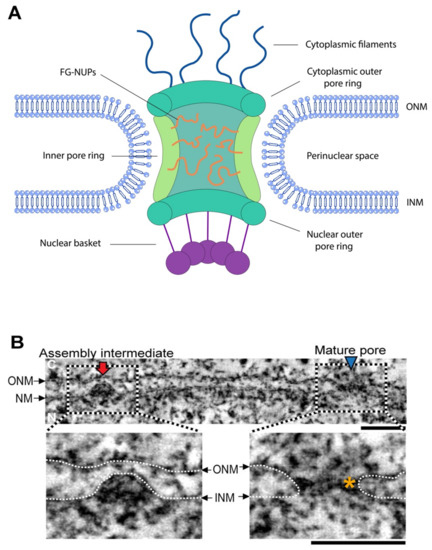
Cells | Free Full-Text | Generating Membrane Curvature at the Nuclear Pore: A Lipid Point of View | HTML

Regioselective Ring Opening of 1,3‐Dioxane‐Type Acetals in Carbohydrates - Janssens - 2018 - European Journal of Organic Chemistry - Wiley Online Library

Synthesis of the seco‐Limonoid BCD Ring System Identifies a Hsp90 Chaperon Machinery (p23) Inhibitor - Pinkerton - 2019 - Chemistry – A European Journal - Wiley Online Library

Beaded Halo Monogram Engravable 925 Sterling Silver Oval Signet Ring for Women for Teen Split Shank Band - Walmart.com

The Timing and Frequency of Pulmonary Veins Unexcitability Relative to Completion of a Wide Area Circumferential Ablation Line for Pulmonary Vein Isolation - ScienceDirect

AC, DC, and hybrid control strategies for smart microgrid application: A review - Sahoo - 2021 - International Transactions on Electrical Energy Systems - Wiley Online Library